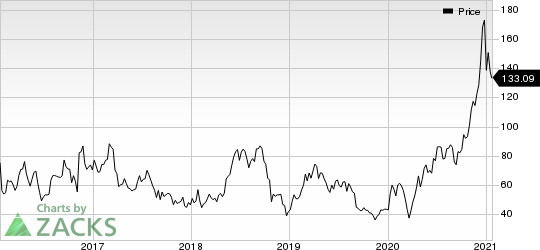
Navigating the Complex Market Landscape of Rare Genetic Disorders: A Deep Dive into Hereditary Angioedema, Wilson Disease, Menkes Disease, and Alpha-1 Antitrypsin Deficiency (AATD) | DelveInsight
www.ultragenyx.com Transforming good science into great medicine for rare genetic diseases February 17, 2022 Dear Members and Fr

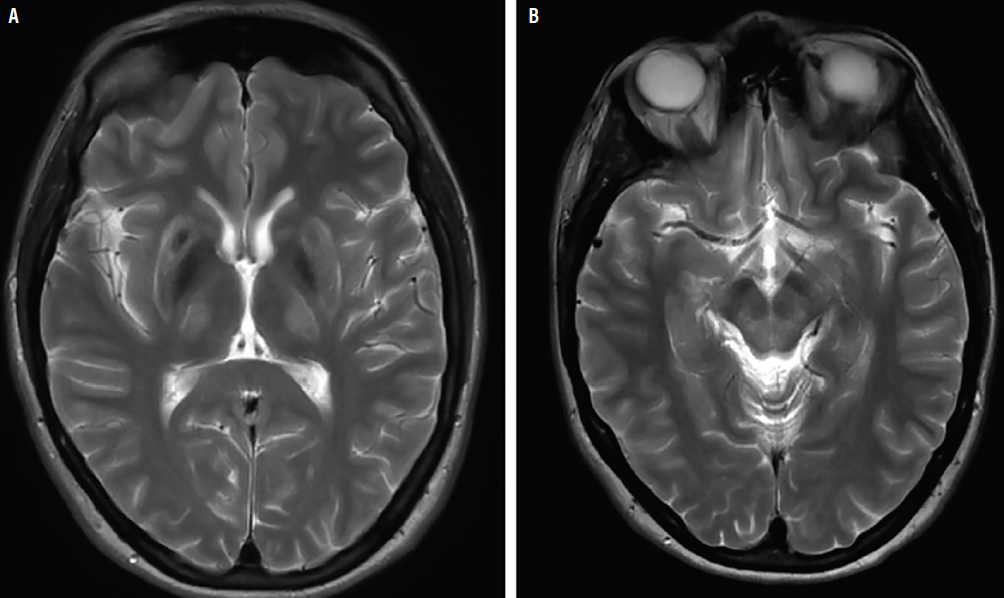
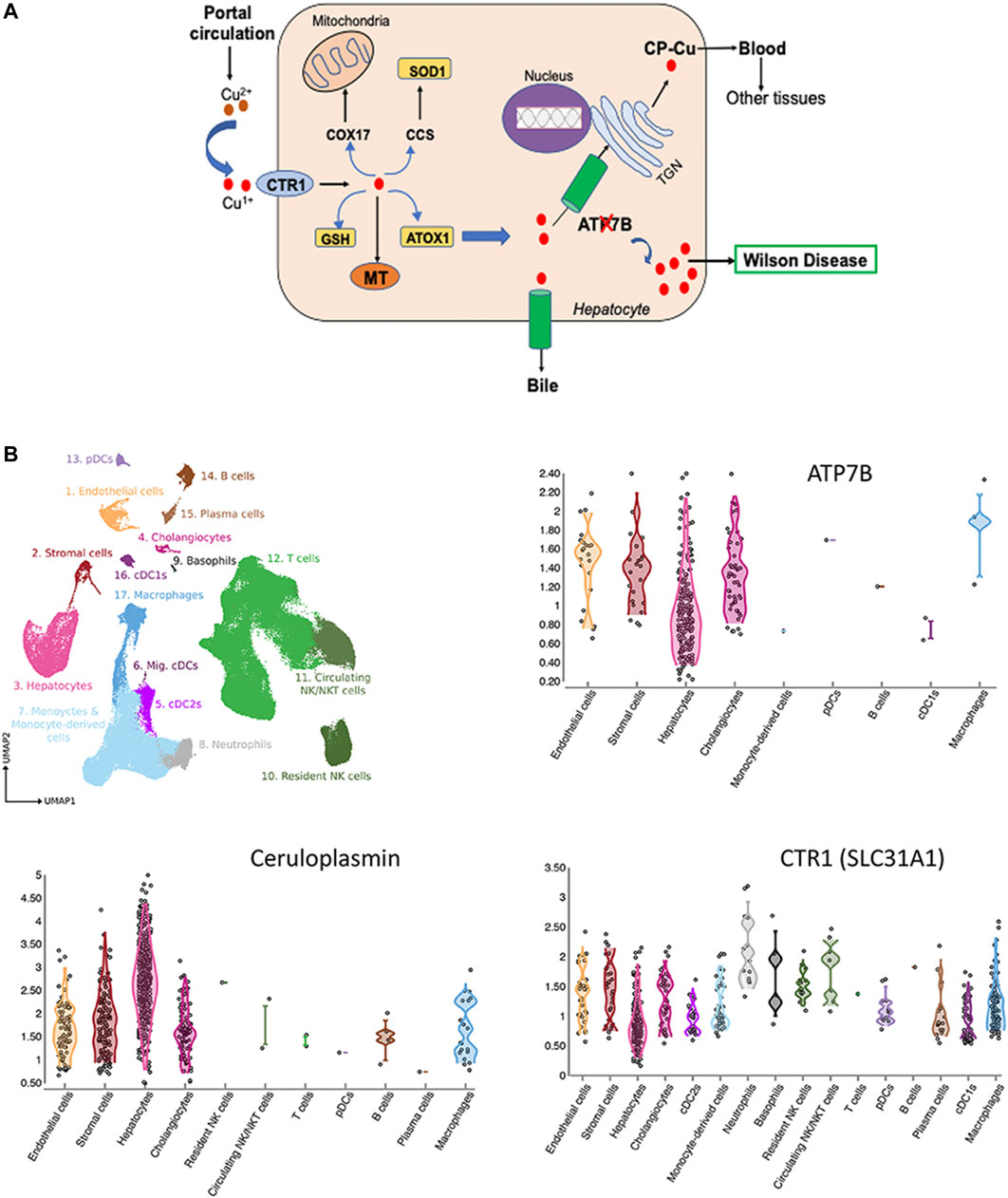
Investigation and management of Wilson's disease: a practical guide from the British Association for the Study of the Liver - The Lancet Gastroenterology & Hepatology

Ultragenyx announces US FDA has granted orphan drug designation to UX701 for the treatment of Wilson Disease